A Review of The Analytical Methods Established for FDA Approved Drugs In 2022
Keywords:
FDA approved drugs, HPLC, LC-MS/MS, Analytical methodsAbstract
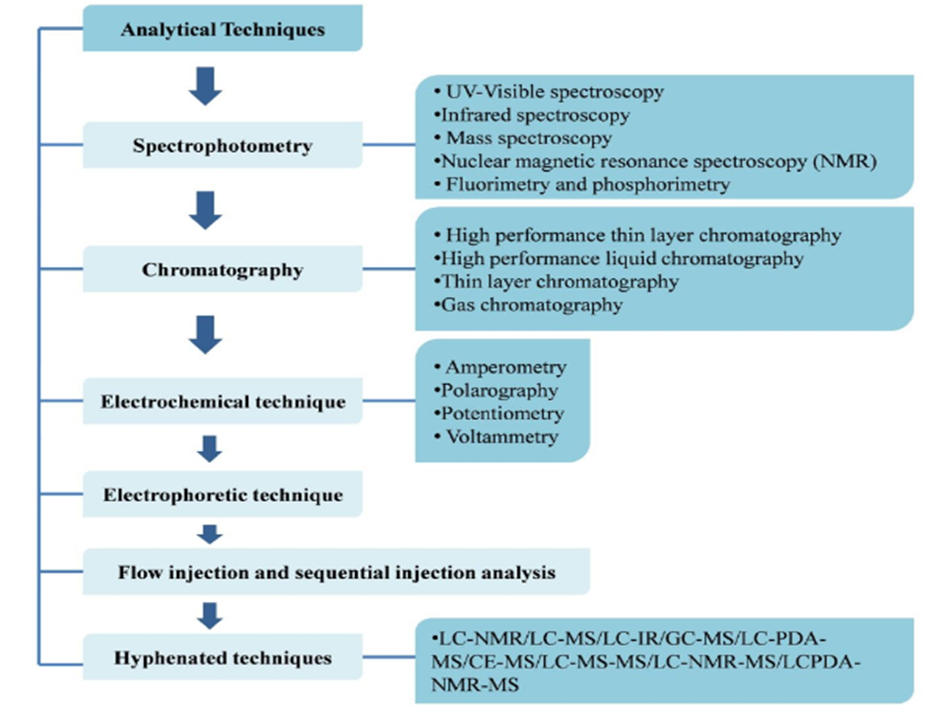
In response to the rising number of diseases, numerous new medications have been developed and approved by the FDA to treat various disorders. Before these drugs can enter the market, they must undergo extensive validation and analytical processes to ensure their purity and reliability. These approaches use a variety of analytical methods to gather information regarding the drugs. This overview covers a variety of analytical procedures, including UV spectrophotometry & chromatography approaches (including HPTLC, HPLC, and Gas Chromatography). Additionally, it discusses hyphenated techniques like LC-MS used in the development of newly approved drugs in 2022. In 2022, significant advancements were made in the refinement of these analytical methods, introducing more precise, rapid, and sensitive techniques for drug evaluation. A notable trend was the increased integration of automation and artificial intelligence in analytical processes, enhancing the accuracy of results and streamlining workflows, thereby facilitating quicker approval timelines for emerging pharmaceuticals.
References
[1] Ravisankar P, Gowthami S, Rao GD (2014) A review on analytical method development 1. Indian J Res Pharm Biotechnol 2(3):1183-95.
[2] Sharma S, Singh N, Ankalgi AD, Rana A, Ashawat MS (2021) Analytical techniques for method development and validation of pharmaceuticals: a review. Journal of Drug Delivery and Therapeutics Modern Trends11:121-30.
[3] Ozkan SA (2018) Analytical method validation: the importance for pharmaceutical analysis. Pharm Sci 24(1):1-2. doi: 10.15171/PS.2018.01.
[4] U.S. Food and Drug Administration (2023).
[5] Ramya K, Theja I, Tejaswi G, Arshiya Md, Sanvi K (2023) A Text Book on Nutraceuticals and Their Determination by Modern Analytical Techniques. https://doi.org/10.22271/int.book.276.
[6] Tang LWT, Chan ECY (2022) Quantification of the irreversible fibroblast growth factor receptor inhibitor futibatinib by UPLC-MS/MS: Application to the metabolic stability assay in human liver microsomes for the estimation of its in vitro hepatic intrinsic clearance. Journal of Pharmaceutical and Biomedical Analysis 214 (114731). https://doi.org/10.1016/j.jpba.2022.114731.
[7] Li H, Ding H, Wang Y, Zhang F (2022) Validated UPLC-MS/MS Method for Determination of Futibatinib and Its Pharmacokinetics in Beagle Dogs. Journal of Chemistry 2022.
[8] Irene AR, Nancy HCL, Alfred HS, Jos HB, Rolf WS (2023) Development and validation of an HPLC–MS/MS method to quantify the KRAS inhibitor adagrasib in mouse plasma and tissue-related matrices. Biomed Chromatogr. 37(11):e5720. doi: 10.1002/bmc.5720.
[9] Kamma HS, Panchumarthy R, Sathish KK, Babu PS (2024) Application of newly developed and validated lc-ms/ms method for pharmacokinetic study of adagrasib and pembrolizumab simultaneously in rat plasma. Journal of Chromatography B 124171. https://doi.org/10.1016/j.jchromb.2024.124171.
[10] Paul DK, Yvo MB, Judith LG, Leo MLS, Lizza ELH, Sander C, Robin MJMG (2023) Validated extended multiplexed LC-MS/MS assay for the quantification of adagrasib and sotorasib in human plasma, together with four additional SMIs. Journal of Chromatography 1231: 123918.
[11] Beludari, Mahammad I, Chedela H, Lingareddy S, China B, Karimulla S (2024) Development and validation of a stability-indicating analytical method for simultaneous determination of sodium phenylbutyrate and taurursodiol in bulk and formulation using reverse phase ultra-performance liquid chromatography. Egyptian Pharmaceutical Journal 23(2):p 264-271. DOI: 10.4103/epj.epj_295_23.
[12] Mahesh P, Haque MA, Salman BI, Belal TS, Ibrahim AE, Deeb SE (2023) Fast and Sensitive Bioanalytical Method for the Determination of Deucravacitinib in Human Plasma Using HPLC-MS/MS: Application and Greenness Evaluation. Molecules 28(14), 5471. https://doi.org/10.3390/molecules28145471
[13] Golla VM, Khemchandani R, Chaganti S, Samanthula G () Separation and characterization of hydrolytic degradation product of deucravacitinib by validated high‐performance liquid chromatography method and liquid chromatography‐mass spectrometry. Separation Science Plus 7(5). DOI:10.1002/sscp.202300180.
[14] Chavva RR, Gosu NR (2024) Developed and Validated for the Estimation of Tapinarof in Topical Formulation and Active Pharmaceutical Ingredients. Journal of AOAC International 107(1), 46-51.
[15] Mansour NM, El-Masry AA, El-Sherbiny DT, Moustafa MA (2024) White analytical insight for sensitive fluorescent determination of semaglutide and tirzepatide in pharmaceuticals and biological matrices. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 313, 124159.
[16] Salva C, Galla R (2024) The development and validation of a stability indicating RP-UPLC method for the simultaneous estimation of clarithromycin, amoxicillin, and vonoprazan in a physical mixture. J Appl Pharm Sci 14(05):193–202. http://doi.org/10.7324/JAPS.2024.165836.
[17] Anusha K, Sowjanya G (2024) A Reliable RP-UPLC-TUV Method For Simultaneous Estimation Of Clarithromycin, Amoxicillin, And Vonoprazan In Co-Packed Pharmaceutical Dosage Forms: Method Development And Validation With Stability Indicating Properties. Int J App Pharm 16(2): 351-357.
[18] Wu YM, Tang PF, Qian J, Xiao ZX (2023) Development of a Rapid Lc-Ms/Ms Method and its Application for the Pharmacokinetic Analysis of Pacritinib in Rats. http://dx.doi.org/10.2139/ssrn.4534015
[19] SM CK, Jagadhathri V, Harshitha P, Sindhuja G, Sumathi B (2024) Development and validation of Abrocitinib In RP-HPLC. Frontier Journal of Pharmaceutical Sciences and Research 7(1): 14-18.
[20] Tachet J, Versace F, Mercier T, Buclin T, Decosterd LA, Choong E, Girardin FR (2023) Development and validation of a multiplex HPLC-MS/MS assay for the monitoring of JAK inhibitors in patient plasma. Journal of Chromatography B 1230: 123917.
[21] Husnain F, Mohammed Y, Mohamed K (2023) Estimation and validation of abrocitinib dosage form and in bulk drug by spectrophotometric method. Asian Journal of Research in Chemistry 16(3):230-4. doi: 10.52711/0974-4150.2023.00037
[22] Susmitha A, Eri GK, Rajitha G (2024). Utilizing central composite design (CCD) in the optimization of new HPLC-PDA method for the assay of daridorexant: Application to the analysis in bulk and tablet dosage forms. Analytical Chemistry Letters, 1-20.
Downloads
Published
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Articles in this journal are licensed under the Creative Commons Attribution-NonCommercial 4.0 International License. This license permits others to copy, distribute, and adapt the work, provided it is for non-commercial purposes, and the original author and source are properly credited.




